Background. Chronic myelomonocytic leukemia (CMML) is a rare myeloid neoplasm that shares both dysplastic and proliferative features and includes patients with highly heterogeneous clinical manifestations and prognosis. Despite known co-mutation patterns that are suggesting of a CMML diagnosis such as TET2 and SRSF2 mutations, the current diagnosis and classification schemes rely solely on morphological criteria that fail to capture the genomic heterogeneity of the disease. Here we perform an unsupervised analysis using only genomic features to generate a novel classification schema of CMML patients with unique clinical features.
Methods. A cohort of 2,471 CMML patients defined according to WHO 2016 criteria from European and US centers was analyzed. Bayesian network analysis and hierarchical Dirichlet processes were used to identify genomic associations and subgroups as a basis to define a molecular classification of CMML. Bayesian networks allow to infer the structure of conditional dependencies among mutations. Dirichlet processes were applied to define clusters capturing broad dependencies among all gene mutations and cytogenetic abnormalities. Multivariate logistic regression analysis was applied to compare clinical and hematological characteristics among different groups. ( JCO 2021 39:11, 1223-1233)
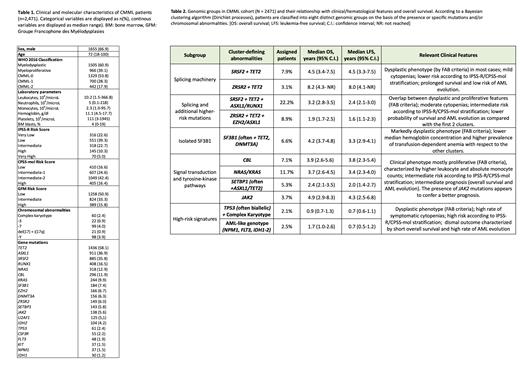
Results. Baseline clinical and molecular characteristics are summarized in Table 1. We identified 11 clusters defined according to specific genomic features (Table 2). Two clusters were primarily defined by the presence of mutations in splicing genes SRSF2 and ZRSR2, frequently associated with TET2 mutations. Patients assigned to these clusters showed myelodysplastic features according to French American British (FAB) criteria, had a low rate of peripheral blood cytopenias, and low blast counts. Additionally, two clusters were characterized by the same mutations in splicing genes (i.e., SRSF2 or ZRSR2 with associated TET2), together with co-mutations in genes that correlate with a more aggressive phenotype ( RUNX1, ASXL1, EZH2). These groups showed overlapping myelodysplastic and myeloproliferative features by FAB, intermediate-risk per prognostic scores, and decreased survival as compared with the first 2 clusters. A distinct cluster was characterized by SF3B1 gene mutations (frequently associated with TET2 or DNMT3A). The SF3B1 cluster was marked by a dysplastic phenotype and more severe anemia (with a high rate of transfusion-dependence) with respect to the other genomic groups. Four clusters (27.8% of the overall population) were characterized by the presence of mutations in signal transduction and tyrosine kinase pathways genes ( NRAS/KRAS, CBL, JAK2 and SETBP1 often associated with ASXL1/TET2, respectively). Patients within these groups showed a mostly proliferative phenotype and intermediate survival. The presence of JAK2 mutations appeared to confer a better prognosis compared with other proliferative clusters. A minority of patients (4.6%) were assigned to two high-risk clusters, defined by TP53 mutations (often biallelic) with complex karyotype, and AML-like mutations ( NPM1, IDH1/2, FLT3), respectively. Patients in these clusters were predominantly dysplastic by FAB, had a high prevalence of severe cytopenias, higher blast counts, and a dismal outcome. Finally, we identified a cluster of patients defined by the absence of gene mutations and chromosomal abnormalities (4.5%). The majority of patients assigned to this cluster were characterized by a dysplastic phenotype, had low absolute monocyte counts, and favorable outcome. A subset of patients (18.9%) could not be assigned to a specified cluster in this preliminary analysis. An independent validation of this molecular classification is currently being conducted on a US prospective cohort (n=650).
Conclusion. Our results provide proof of concept for a molecular classification of CMML based on the identification of homogeneous genomic signatures and associated clinical features. 15% of patients showed clear overlapping features with other myeloid neoplasms, thus providing the rational to refine the boundaries among different clinical entities. A molecular classification of CMML may provide a basis to define a next-generation prognostic tool and improve clinical decision-making.
Disclosures
Hunter:Sierra Oncology: Membership on an entity's Board of Directors or advisory committees. Meggendorfer:MLL Munich Leukemia Laboratory: Current Employment. Jerez:GILEAD: Research Funding; Novartis: Consultancy; Astrazeneca: Research Funding; BMS: Consultancy. Díaz-Beyá:Bristol Myers Squibb: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria; Astellas: Consultancy, Honoraria; Jazz Pharma: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Novartis: Consultancy, Honoraria. Passamonti:Roche: Consultancy; Abbvie: Consultancy, Honoraria; Novartis, GSK, Bristol Myers Squibb, Celgene, Sierra Oncology, AbbVie, Janssen, Roche, AOP Orphan, Karyopharm, Kyowa Kirin, MEI, Sumitomo: Honoraria; BMS: Consultancy, Honoraria, Research Funding. Santoro:Sanofi: Consultancy; Incyte: Consultancy; BMS: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Servier: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Pfizer: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Gilead: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Bayer: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Abbvie: Speakers Bureau; Roche: Speakers Bureau; Takeda: Speakers Bureau; Merck MSD: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Eisai: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Amgen: Speakers Bureau; Celgene (BMS): Speakers Bureau; AstraZeneca: Speakers Bureau; Eli Lilly: Speakers Bureau; Sandoz: Speakers Bureau; Novartis: Speakers Bureau; Arqule: Other. Kern:MLL Munich Leukemia Laboratory: Current Employment, Other: Equity Ownership. Platzbecker:Syros: Consultancy, Honoraria, Research Funding; Takeda: Consultancy, Honoraria, Research Funding; Merck: Research Funding; Servier: Consultancy, Honoraria, Research Funding; Fibrogen: Research Funding; MDS Foundation: Membership on an entity's Board of Directors or advisory committees; Roche: Research Funding; Janssen Biotech: Consultancy, Research Funding; Geron: Consultancy, Research Funding; Curis: Consultancy, Research Funding; Celgene: Honoraria; Silence Therapeutics: Consultancy, Honoraria, Research Funding; Jazz: Consultancy, Honoraria, Research Funding; AbbVie: Consultancy; Novartis: Consultancy, Honoraria, Research Funding; Bristol Myers Squibb: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel support; medical writing support, Research Funding; Amgen: Consultancy, Research Funding; BeiGene: Research Funding; BMS: Research Funding. Diez-Campelo:BMS/Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Advisory board fees; Gilead Sciences: Other: Travel expense reimbursement; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; GSK: Consultancy, Membership on an entity's Board of Directors or advisory committees. Sallman:AbbVie, Affimed Gmbh, Gilead, Incyte, Intellisphere, LLC, Molecular Partners AG, PGEN Therapeutics, Inc., Takeda, Zentalis; Advisory board for AvenCell, BlueBird Bio, BMS, Intellia, Jasper Therapeutics, Kite, Magenta Therapeutics, NKARTA, Novartis, Orbita: Consultancy; Aprea, Jazz: Research Funding. Fenaux:French MDS Group: Honoraria; Novartis: Consultancy, Honoraria, Research Funding; Janssen: Consultancy, Honoraria, Research Funding; AbbVie: Consultancy, Honoraria, Research Funding; Bristol Myers Squibb: Consultancy, Honoraria, Research Funding; Jazz: Consultancy, Honoraria, Research Funding. Zeidan:Foran: Consultancy, Research Funding; Servier: Consultancy, Honoraria; Daiichi Sankyo: Consultancy, Honoraria; Taiho: Consultancy, Honoraria; Astellas: Consultancy, Honoraria; Boehringer-Ingelheim: Consultancy, Honoraria; Ionis: Consultancy, Honoraria; Syndax: Consultancy, Honoraria; Kura: Consultancy, Honoraria; Syros: Consultancy, Honoraria; BioCryst: Consultancy, Honoraria; Shattuck Labs: Research Funding; Chiesi: Consultancy, Honoraria; Lox Oncology: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Otsuka: Consultancy, Honoraria; Mendus: Consultancy, Honoraria; Orum: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; Epizyme: Consultancy, Honoraria; AbbVie: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria; Gilead: Consultancy, Honoraria; ALX Oncology: Consultancy, Honoraria; Schrödinger: Consultancy, Honoraria; Notable: Consultancy, Honoraria; Incyte: Consultancy, Honoraria; Agios: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Geron: Consultancy, Honoraria; Jazz: Consultancy, Honoraria; Seattle Genetics: Consultancy, Honoraria; BeyondSpring: Consultancy, Honoraria; Regeneron: Consultancy, Honoraria; Tyme: Consultancy, Honoraria; Zentalis: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Celgene/BMS: Consultancy, Honoraria; Genentech: Consultancy, Honoraria; Astex: Research Funding. Komrokji:BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; AbbVie, CTI biopharma, Jazz, Pharma Essentia, Servio: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Geron: Consultancy; Rigel, Taiho, DSI: Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees. Santini:BMS, Abbvie, Geron, Gilead, CTI, Otsuka, servier, janssen, Syros: Membership on an entity's Board of Directors or advisory committees. Haferlach:MLL Munich Leukemia Laboratory: Current Employment, Other: Equity Ownership. Padron:Kura: Research Funding; Abbvie: Membership on an entity's Board of Directors or advisory committees; BMS: Research Funding; Incyte: Research Funding; CTI: Membership on an entity's Board of Directors or advisory committees; Pharmaessentia: Membership on an entity's Board of Directors or advisory committees; Gillead: Membership on an entity's Board of Directors or advisory committees. Della Porta:Bristol Myers Squibb: Honoraria, Membership on an entity's Board of Directors or advisory committees.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal